Product Info Summary
| SKU: | PA1013-1 |
|---|---|
| Size: | 100 μg/vial |
| Reactive Species: | Human, Mouse, Rat |
| Host: | Rabbit |
| Application: | Flow Cytometry, IP, IF, IHC, ICC, WB |
Customers Who Bought This Also Bought
Product info
Product Name
Anti-Apoptosis regulator BAX Bax Antibody Picoband®
SKU/Catalog Number
PA1013-1
BA0315-2 is an alternative SKU for this antibody, used in previous lots.
Size
100 μg/vial
Form
Lyophilized
Description
Boster Bio Anti-Apoptosis regulator BAX Bax Antibody catalog # PA1013-1. Tested in Flow Cytometry, IP, ICC/IF, IHC, WB applications. This antibody reacts with Human, Mouse, Rat. The brand Picoband indicates this is a premium antibody that guarantees superior quality, high affinity, and strong signals with minimal background in Western blot applications. Only our best-performing antibodies are designated as Picoband, ensuring unmatched performance.
Storage & Handling
Store at -20˚C for one year from date of receipt. After reconstitution, at 4˚C for one month. It can also be aliquotted and stored frozen at -20˚C for six months. Avoid repeated freeze-thaw cycles.
Cite This Product
Anti-Apoptosis regulator BAX Bax Antibody Picoband® (Boster Biological Technology, Pleasanton CA, USA, Catalog # PA1013-1)
Host
Rabbit
Contents
Each vial contains 4 mg Trehalose, 0.9 mg NaCl and 0.2 mg Na2HPO4.
Clonality
Polyclonal
Isotype
Rabbit IgG
Immunogen
A synthetic peptide corresponding to a sequence at the N-terminus of human Bax, different from the related mouse and rat sequences by one amino acid.
Cross-reactivity
No cross-reactivity with other proteins
Reactive Species
PA1013-1 is reactive to BAX in Human, Mouse, Rat
Observed Molecular Weight
21 kDa
Calculated molecular weight
21.2 kDa
Background of BAX
Apoptosis regulator BAX, also known as bcl-2-like protein 4, is a protein that in humans is encoded by the BAX gene. The protein encoded by this gene belongs to the BCL2 protein family. BCL2 family members form hetero- or homodimers and act as anti- or pro-apoptotic regulators that are involved in a wide variety of cellular activities. This protein forms a heterodimer with BCL2, and functions as an apoptotic activator. Additionally, this protein is reported to interact with, and increase the opening of, the mitochondrial voltage-dependent anion channel (VDAC), which leads to the loss in membrane potential and the release of cytochrome c. The expression of this gene is regulated by the tumor suppressor P53 and has been shown to be involved in P53-mediated apoptosis. Multiple alternatively spliced transcript variants, which encode different isoforms, have been reported for this gene.
Antibody Validation
Boster validates all antibodies on WB, IHC, ICC, Immunofluorescence, and ELISA with known positive control and negative samples to ensure specificity and high affinity, including thorough antibody incubations.
Application & Images
Applications
PA1013-1 is guaranteed for Flow Cytometry, IP, IF, IHC, ICC, WB Boster Guarantee
Recommend Dilution
| Application | Dilution | Species |
|---|---|---|
| Western blot | 0.1-0.5μg/ml | Human, Mouse, Rat |
| Immunohistochemistry (Paraffin-embedded Section) | 2-5μg/ml | Human |
| Immunocytochemistry/Immunofluorescence | 5 μg/ml | Human |
| Immunoprecipitation | 0.5-2 μg/ml | Human |
| Flow Cytometry (Fixed) | 1-3μg/1x106 cells | Human |
Tested application
Suggested blocking solution with 5% non-fat milk or BSA; (*)Recommended protein loading: 20-40 µg per lane
Use TE buffer pH 9.0 for antigen retrieval; (*) citrate buffer pH 6.0 is an alternative.
Validation Images & Assay Conditions
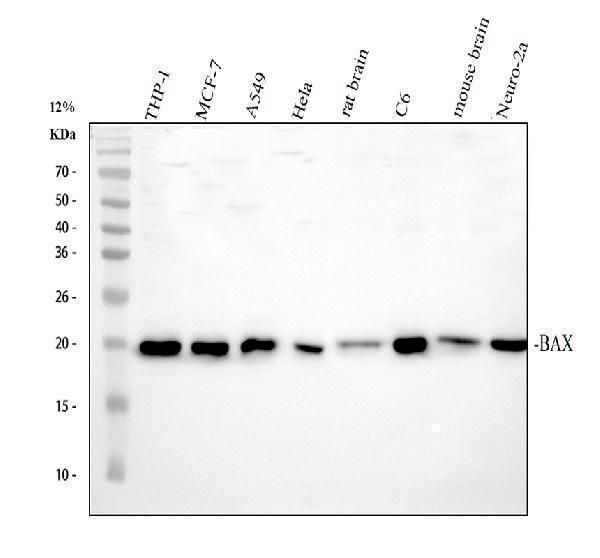
Click image to see more details
Western blot analysis of BAX using anti-BAX antibody (PA1013-1).
Electrophoresis was performed on a 12% SDS-PAGE gel at 80V (Stacking gel) / 120V (Resolving gel) for 2 hours. The sample well of each lane was loaded with 30 ug of sample under reducing conditions.
Lane 1: human THP-1 whole cell lysates,
Lane 2: human MCF-7 whole cell lysates,
Lane 3: human A549 whole cell lysates,
Lane 4: human Hela whole cell lysates,
Lane 5: rat brain tissue lysates,
Lane 6: rat C6 whole cell lysates,
Lane 7: mouse brain tissue lysates,
Lane 8: mouse Neuro-2a whole cell lysates.
After electrophoresis, proteins were transferred to a nitrocellulose membrane at 150 mA for 50-90 minutes. Blocked the membrane with 5% non-fat milk/TBS for 1.5 hour at RT. The membrane was incubated with rabbit anti-BAX antigen affinity purified polyclonal antibody (PA1013-1) at 0.5 μg/mL overnight at 4°C, then washed with TBS-0.1%Tween 3 times with 5 minutes each and probed with a goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:5000 for 1.5 hour at RT. The signal is developed using an ECL Plus Western Blotting Substrate (Catalog # AR1196-200) with Tanon 5200 system. A specific band was detected for BAX at approximately 21 kDa. The expected band size for BAX is at 21 kDa.
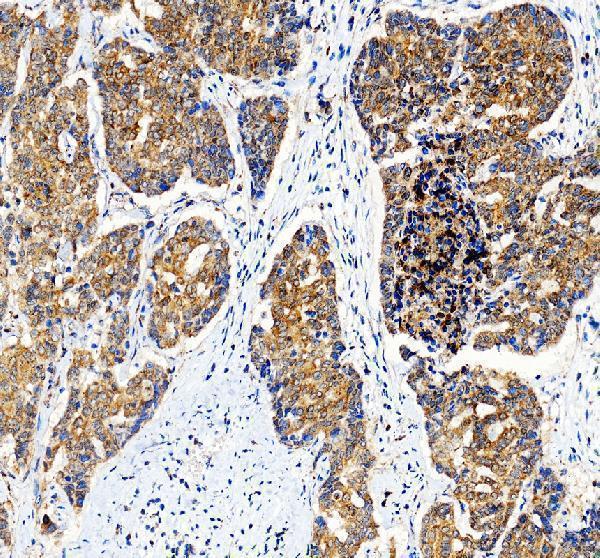
Click image to see more details
IHC analysis of BAX using anti-BAX antibody (PA1013-1).
BAX was detected in a paraffin-embedded section of human liver cancer tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-BAX Antibody (PA1013-1) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
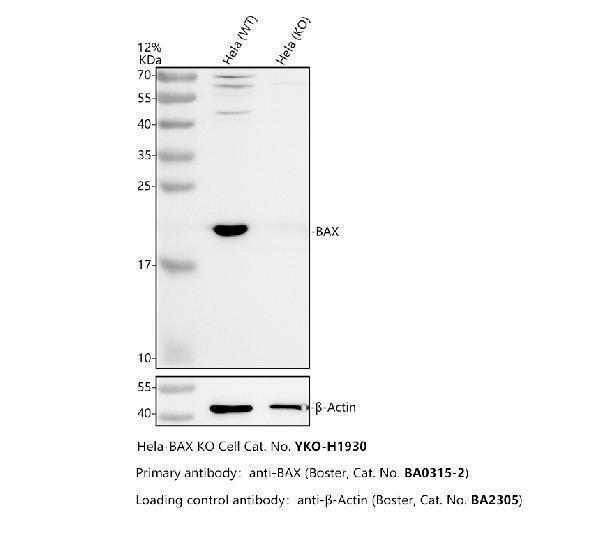
Click image to see more details
Western blot analysis of BAX using anti-BAX antibody (PA1013-1).
Electrophoresis was performed on a 12% SDS-PAGE gel at 80V (Stacking gel) / 120V (Resolving gel) for 2 hours. The sample well of each lane was loaded with 30 ug of sample under reducing conditions.
Lane 1: human Hela- WT whole cell lysates,
Lane 2: human Hela-BAX KO whole cell lysates.
After electrophoresis, proteins were transferred to a nitrocellulose membrane at 150 mA for 50-90 minutes. Blocked the membrane with 5% non-fat milk/TBS for 1.5 hour at RT. The membrane was incubated with rabbit anti-BAX antigen affinity purified polyclonal antibody (PA1013-1) at 0.5 μg/mL overnight at 4°C, then washed with TBS-0.1%Tween 3 times with 5 minutes each and probed with a goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:5000 for 1.5 hour at RT. The signal is developed using an ECL Plus Western Blotting Substrate (Catalog # AR1196-200) with Tanon 5200 system. A specific band was detected for BAX at approximately 21 kDa. The expected band size for BAX is at 21 kDa.

Click image to see more details
IHC analysis of BAX using anti-BAX antibody (PA1013-1).
BAX was detected in a paraffin-embedded section of human tonsil tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-BAX Antibody (PA1013-1) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
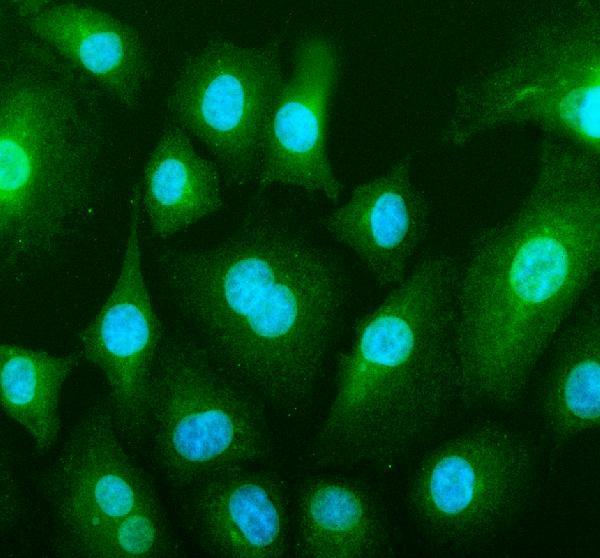
Click image to see more details
IF analysis of BAX using anti-BAX antibody (PA1013-1).
BAX was detected in an immunocytochemical section of A549 cells. Enzyme antigen retrieval was performed using IHC enzyme antigen retrieval reagent (AR0022) for 15 mins. The cells were blocked with 10% goat serum. And then incubated with 5 μg/mL rabbit anti-BAX Antibody (PA1013-1) overnight at 4°C. Fluoro488 Conjugated Goat Anti-Rabbit IgG (BA1127) was used as secondary antibody at 1:500 dilution and incubated for 30 minutes at 37°C. The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.
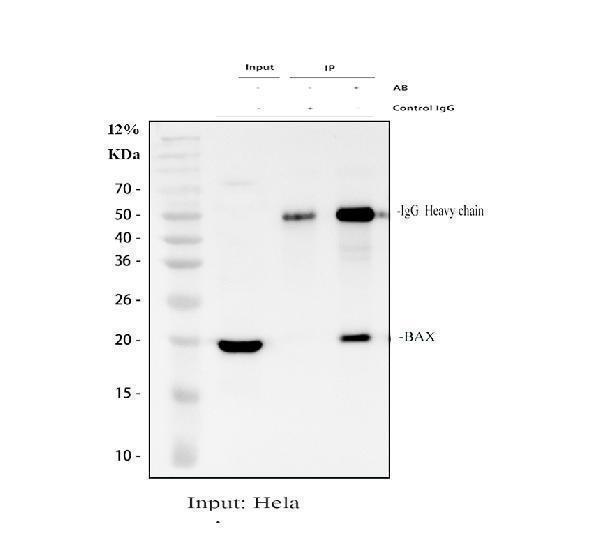
Click image to see more details
Immunoprecipitating (IP) BAX in Hela whole cell lysate.
Western blot analysis of BAX using anti-BAX antibody (PA1013-1);
Lane 1: Hela whole cell lysates (30ug);
Lane 2: Rabbit control IgG instead of anti-BAX antibody in Hela whole cell lysate;
Lane 3: anti-BAX antibody (2μg) + Hela whole cell lysate (500μg).
After electrophoresis, proteins were transferred to a membrane. Then the membrane was incubated with rabbit anti-BAX antigen affinity purified polyclonal antibody (PA1013-1) at a dilution of 0.5 μg/mL and probed with a goat anti-rabbit IgG-HRP secondary antibody (Catalog # BA1054). The signal is developed using ECL Plus Western Blotting Substrate (Catalog # AR1196-200). A specific band was detected for BAX at approximately 21 kDa. The expected band size for BAX is at 21 kDa.
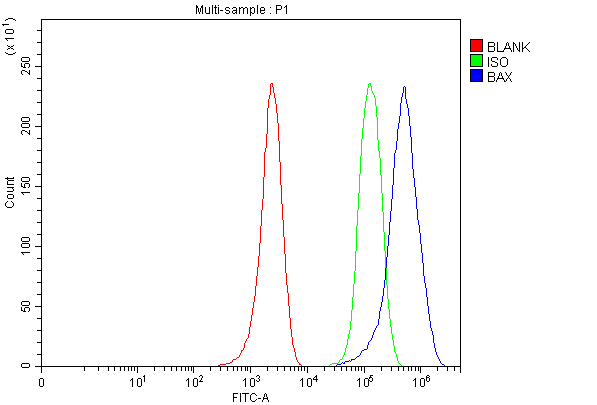
Click image to see more details
Flow Cytometry analysis of MCF-7 cells using anti-BAX antibody (PA1013-1).
Overlay histogram showing MCF-7 cells stained with PA1013-1 (Blue line). To facilitate intracellular staining, cells were fixed with 4% paraformaldehyde and permeabilized with permeabilization buffer. The cells were blocked with 10% normal goat serum. And then incubated with rabbit anti-BAX Antibody (PA1013-1, 1 μg/1x106 cells) for 30 min at 20°C. DyLight®488 conjugated goat anti-rabbit IgG (BA1127, 5-10 μg/1x106 cells) was used as secondary antibody for 30 minutes at 20°C. Isotype control antibody (Green line) was rabbit IgG (1 μg/1x106) used under the same conditions. Unlabelled sample without incubation with primary antibody and secondary antibody (Red line) was used as a blank control.
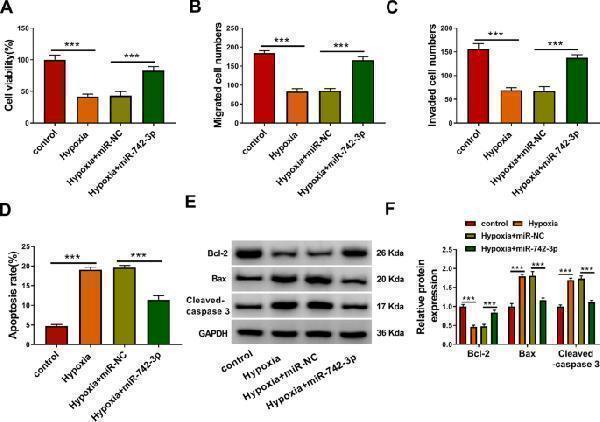
Click image to see more details
MiR-742-3p relieved hypoxia-induced H9c2 cell injury. H9c2 cells were transfected with or without miR-NC (50 nM) or miR-742-3p mimic (50 nM), and then treated with hypoxia. Untreated H9c2 cells were used as control. Cell viability, migrated and invaded cell numbers, and cell apoptosis rate were measured using CCK8 assay ( A ), transwell assay ( B , C ) and flow cytometry ( D ). E , F The protein levels of Bcl-2, Bax and Cleaved-caspase 3 were determined using WB analysis. All experiments were repeated three times. *** P < 0.001
Index in PubMed under a CC BY license. PMID: 35346026
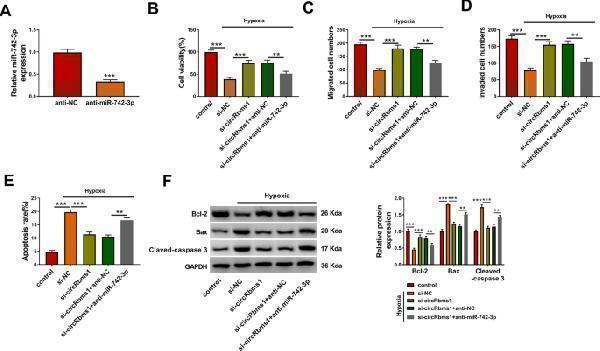
Click image to see more details
Effects of circRbms1 silencing and miR-742-3p inhibitor on hypoxia-induced H9c2 cell injury. A After transfecting with anti-NC (50 nM) or anti- miR-742-3p (50 nM) into H9c2 cells, the expression of miR-742-3p was assessed by qRT-PCR. B–F H9c2 cells were transfected with si-NC (50 nM), si- circRbms1 (50 nM), si- circRbms1 (50 nM) + anti-NC (50 nM), or si- circRbms1 (50 nM) + anti- miR-742-3p (50 nM), and then treated with hypoxia. Untreated H9c2 cells were used as control. CCK8 assay ( B ), transwell assay ( C , D ), and flow cytometry ( E ) were employed to examine cell viability, migrated and invaded cell numbers, and cell apoptosis rate, respectively. F WB analysis was used to test the protein levels of Bcl-2, Bax, and Cleaved-caspase 3. All experiments were repeated three times. ** P < 0.01, *** P < 0.001
Index in PubMed under a CC BY license. PMID: 35346026
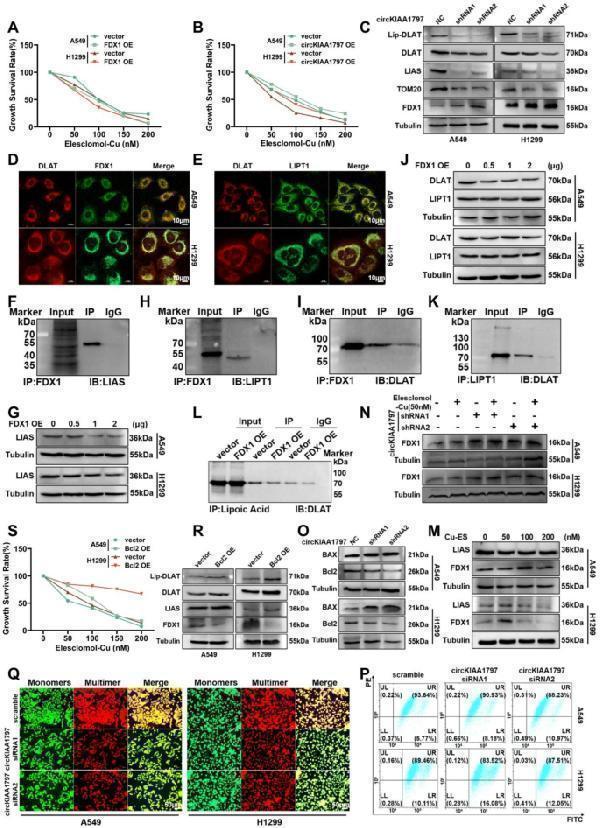
Click image to see more details
Exploration of the mechanism of cuproptosis induction. ( A ) After the transient overexpression of FDX1, a CCK-8 assay was used to detect changes in cellular resistance to cuproptosis. ( B ) After the transient overexpression of circKIAA1797, a CCK-8 assay was performed to detect changes in cellular resistance to cuproptosis. ( C ) Western blot analysis of Lip-DLAT, DLAT, LIAS, TOM20, and FDX1 protein expression after the stable silencing of circKIAA1797. ( D ) Immunofluorescence staining showed the colocalisation of the DLAT and FDX1 proteins. ( E ) Immunofluorescence staining was performed to detect the colocalisation of DLAT with the LIPT1 protein. ( F ) Western blot results of the Co-IP assay used to detect FDX1 binding to LIAS. ( G ) Western blot analysis of LIAS protein expression levels after the gradient overexpression of FDX1. ( H ) Western blot results of the Co-IP assay for detecting FDX1 binding to LIPT1. ( I ) Western blot results of Co-IP experiments for detecting FDX1 binding to DLAT. ( J ) Gradient overexpression of FDX1 followed by Western blot detection of DLAT and LIPT1 protein expression levels. ( K ) Western blot results of the Co-IP assay used to detect LIPT1 binding to DLAT. ( L ) After the overexpression of FDX1, Co-IP experiments were performed to detect changes in the ability of lipoic acid to bind DLAT. ( M ) Cells were treated with 0 nM, 50 nM, 100 nM and 200 nM elesclomol-Cu, and Western blot was performed to examine the protein expression of LIAS and FDX1. ( N ) circKIAA1797 stably transfected cells were treated with 50 nM elesclomol-Cu, and FDX1 protein expression was examined by Western blot. ( O ) Western blot analysis of BAX and Bcl2 protein expression in cell lines with stable circKIAA1797 silencing. ( P ) Flow cytometry detection of changes in the mitochondrial membrane potential after the transient silencing of circKIAA1797. ( Q ) Fluorescence microscopy comparing the changes in JC monomer and multimer levels after the transient silencing of circKIAA1797. ( R ) After the overexpression of Bcl2, Western blot assays were performed to detect Lip-DLAT, DLAT, LIAS, and FDX1 protein expression. ( S ) After the overexpression of Bcl2, a CCK-8 assay was performed to detect changes in cellular resistance to cuproptosis
Index in PubMed under a CC BY license. PMID: 40176113
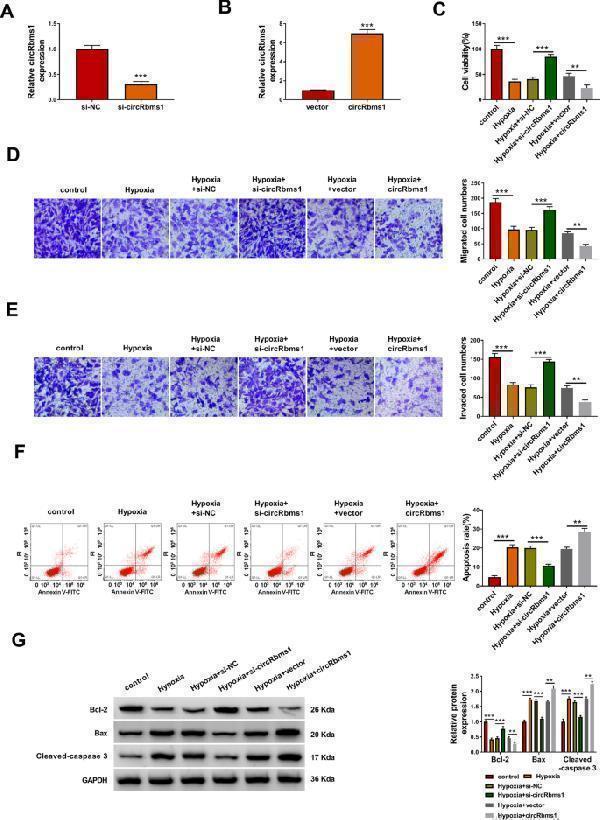
Click image to see more details
Knockdown of circRbms1 alleviated hypoxia-induced H9c2 cell injury. A , B qRT-PCR was used to assess circRbms1 expression to evaluate the transfection efficiency of si-circRbms1 (50 nM) or circRbms1 overexpression vector (4.0 µg) in H9c2 cells. C – G H9c2 cells were transfected with or without si-NC (50 nM), si- circRbms1 (50 nM), vector (4.0 µg) or circRbms1 (4.0 µg), and then treated with hypoxia. Untreated H9c2 cells were used as control. CCK8 assay ( C ), transwell assay ( D , E ) and flow cytometry ( F ) were used to determine cell viability, migrated and invaded cell numbers, and cell apoptosis rate, respectively. G WB analysis was performed to test the protein levels of Bcl-2, Bax, and Cleaved-caspase 3. All experiments were repeated three times. ** P < 0.01, *** P < 0.001
Index in PubMed under a CC BY license. PMID: 35346026
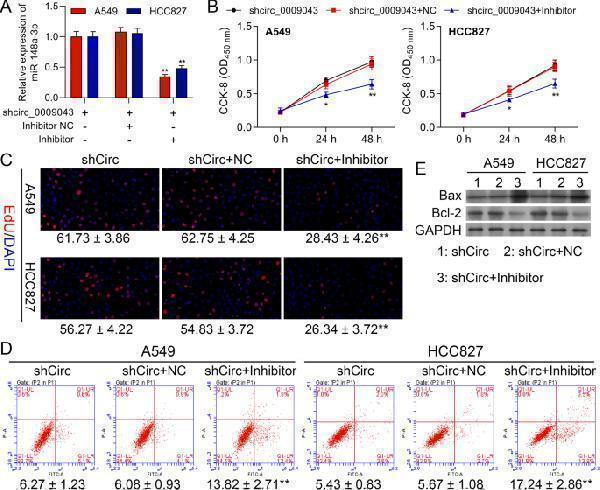
Click image to see more details
MiR-148a-3p Reversed the Regulatory Effect of circ_0009043 on A549 and HCC827 cells. A549 and HCC827 cells were transfected with shCirc, shCirc + NC, or shCirc + miR-148a-3p inhibitor. (A) MiR-148a-3p mRNA levels in A549 and HCC827 were determined with RT-qPCR assay. B Viability in A549 and HCC827 cells at 0, 24, and 48 h. C EdU assay to detect A549 and HCC827 cell proliferation. (D) FSC assay to detect A549 and HCC827 cell apoptosis. E Protein levels of Bax and Bcl-2 in A549 and HCC827 cells with the indicated transfection were determined by western blot. GAPDH is a loading control. Data are presented as mean ± standard deviation. ** P < 0.01; *** P < 0.001
Index in PubMed under a CC BY license. PMID: 35974419
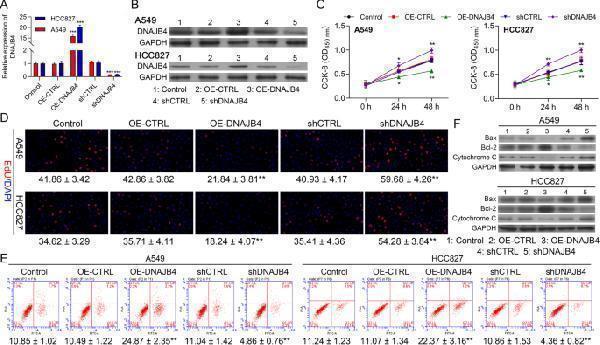
Click image to see more details
Overexpression of DNAJB4 inhibits the proliferation but promotes apoptosis abilities of NSCLC cells. A DNAJB4 mRNA expression in HCC827 and A549 cells transfected with pcDNA4.0 vector, pcDNA4.0- DNAJB4 vector, CTRL‑shRNA, or DNAJB4 ‑shRNA were determined by RT-qPCR. B DNAJB4 protein expression in HCC827 and A549 cells transfected with indicated vectors, which divided into five groups, including Control, OE-CTRL, OE-DNAJB4, shCTRL and shDNAJB4. C CCK8 assay was used to compare the cell proliferation of CTRL, OE-CTRL, OE- DNAJB4, shCTRL and sh DNAJB4 groups in HCC827 and A549 cells. D Edu assay of Control, OE-CTRL, OE- DNAJB4, shCTRL and sh DNAJB4 groups in HCC827 and A549 cells, respectively. E FSC assay to detect cell apoptosis of Control, OE-CTRL, OE- DNAJB4, shCTRL and sh DNAJB4 groups in HCC827 and A549 cells. F Expression level of Bax, Bcl-2 and Cytochrome C in the HCC827 and A549 cells of the 5 groups (Control, OE-CTRL, OE- DNAJB4, shCTRL and sh DNAJB4 groups) were determined by western blotting. Control negative control, OE over expression. Data represent mean values ± SD from three replicates of each sample; ** P < 0.01, *** P < 0.001, **** P < 0.001
Index in PubMed under a CC BY license. PMID: 35974419
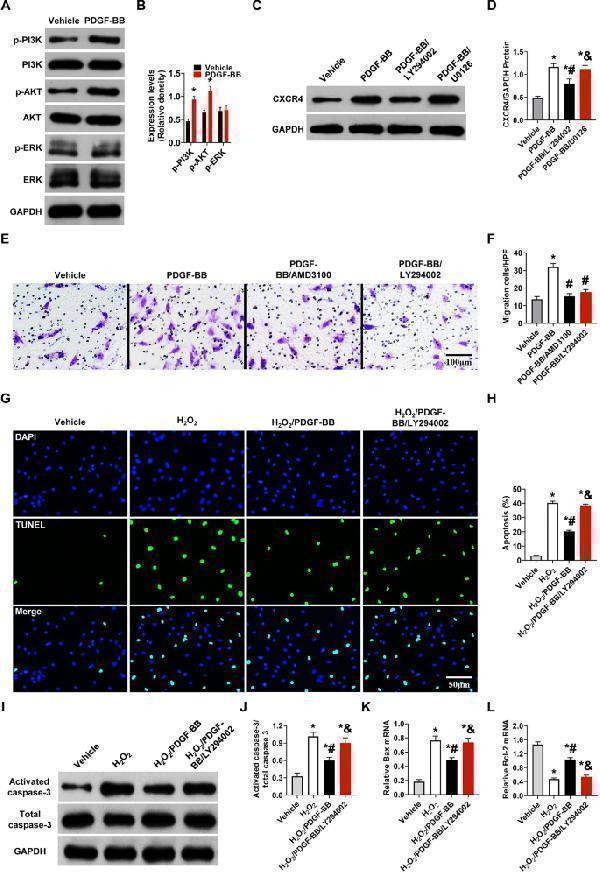
Click image to see more details
PDGF-BB promotes MSC migration and protects MSCs against apoptosis via PI3K/Akt signaling. ( A - B ) Western blot analysis of total and phosphorylated PI3K, Akt, and ERK in MSCs with or without PDGF-BB treatment. PDGF-BB, 50 ng/ml. Values are the mean ± SEM. Significant differences were determined by Student’s t test. N = 6/group. *P < 0.05 vs. vehicle. ( C - D ) Western blot analysis of CXCR4 in MSCs. LY294002, 10 µM, U0126, 10 µM, 2 h before PDGF-BB treatment. Values are the mean ± SEM. Significant differences were determined by using one-way ANOVA. N = 6/group. *p < 0.01 vs. vehicle; # p < 0.01 vs. PDGF-BB; & p < 0.01 vs. PDGF-BB/LY294002. ( E - F ). MSC migratory capacities were evaluated by Transwell assay. LY294002, 50 µM. AMD3100, 44 nM. PDGF-BB, 50 ng/ml. Bar, 100 μm. Values are the mean ± SEM. Significant differences were determined by using one-way ANOVA. N = 6/group. *p < 0.01 vs. vehicle; # p < 0.01 vs. PDGF-BB; & p < 0.01 vs. PDGF-BB/AMD3100. ( G - H ). MSC apoptosis was evaluated by TUNEL assay. Bar, 50 μm. ( I - J ) Western blot analysis of activated caspase-3 in MSCs. ( K - L ) Bax and BCL-2 mRNA expression levels were determined by RT‒PCR in MSCs. H 2 O 2 , 200 µM, 6 h. LY294002, 30 µM. Values are the mean ± SEM. Significant differences were determined by using one-way ANOVA. N = 6/group. *p < 0.01 vs. vehicle; # p < 0.01 vs. H 2 O 2 ; & p < 0.01 vs. H 2 O 2 /PDGF-BB
Index in PubMed under a CC BY license. PMID: 38102643
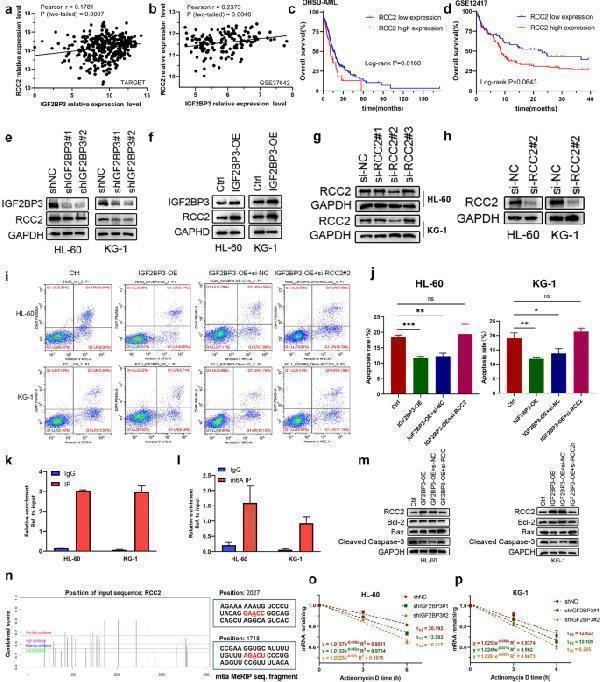
Click image to see more details
IGF2BP3 regulates RCC2 expression in an m6A-dependent manner. a , b RCC2 expression was positively correlated with IGF2BP3 expression in the GSE37642 and TARGET datasets. c , d Kaplan–Meier survival analysis revealed that high RCC2 expression indicated a poor prognosis in AML patients. e , f Protein expression level of RCC2 following knockdown or overexpression of IGF2BP3 in HL-60 and KG-1 cells. g , h The interference efficiency of the siRNAs was evaluated to confirm the feasibility of the siRNAs, and si-RCC2#2 was found to be effective in reducing RCC2 expression. i , j Apoptosis was detected by flow cytometry. RCC2 deficiency promoted the induction of apoptosis by IGF2BP3 overexpression. k The mRNA of RCC2 was enriched by the anti-IGF2BP3 antibody compared to IgG in the HL-60 and KG-1 cell lines. l The mRNA of RCC2 was enriched by the m6A-specific antibody compared to IgG in the HL-60 and KG-1 cell lines. m Overexpression of IGF2BP3 restored the increases in the levels of proapoptotic proteins (Bax and cleaved Caspase 3) caused by silencing RCC2, and the level of the antiapoptotic protein Bcl-2 was slightly decreased. n The potential m6A sites in RCC2 were predicted by SRAMP. The different colored lines indicate different confidence levels. o , p Loss of IGF2BP3 reduced RCC2 stability in HL-60 and KG-1 cells. Transfected cells were treated with 5 µg/ml actinomycin D for 0 h, 3 h, or 6 hours prior to RNA extraction. * P < 0.05; ** P < 0.01; *** P < 0.001.
Index in PubMed under a CC BY license. PMID: 35217832
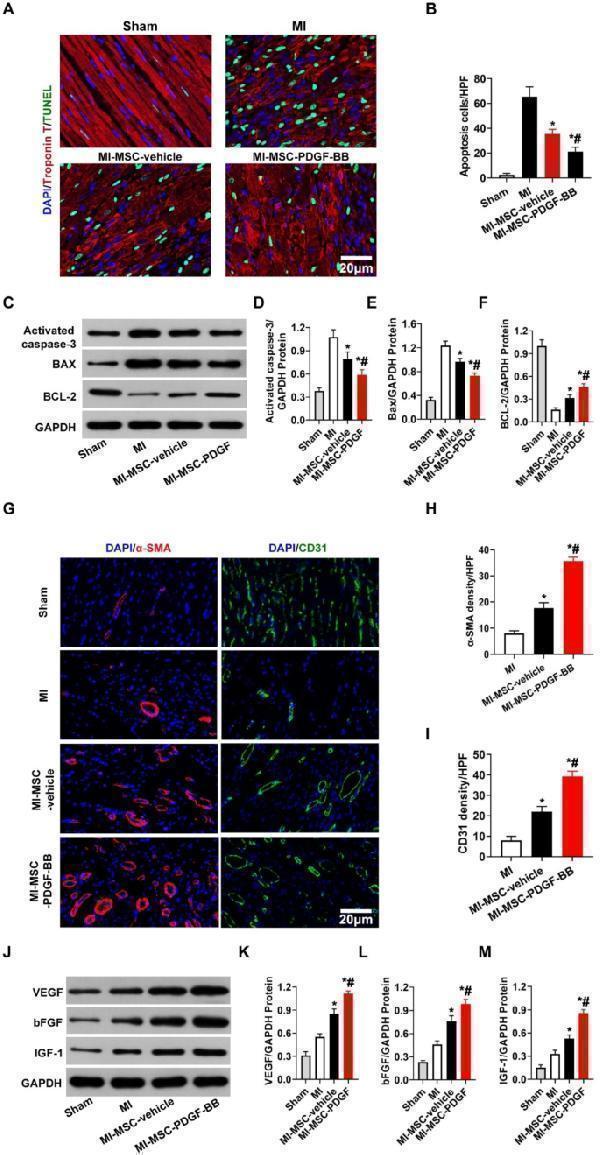
Click image to see more details
Transplantation of PDGF-BB-primed MSCs via UTMD reduces cardiomyocyte apoptosis and improves angiogenesis in rat hearts post-MI. ( A ) Representative images of TUNEL-positive cardiomyocytes in the ischemic area 30 days after MI. Apoptotic nuclei were identified as TUNEL positive (green fluorescence), and total nuclei were identified by DAPI counterstaining (blue fluorescence). Myocardium was stained using a monoclonal antibody against cardiac troponin I (red fluorescent). Bar, 20 μm. ( B ). Quantification of TUNEL-positive cardiomyocytes. ( C - F ) Western blotting of activated caspase 3, Bax, and BCL-2 in the ischemic heart. GAPDH was used as a loading control. ( G ) Representative images of CD31 staining and α-SMA staining in the ischemic hearts of rats 30 days post-MI. Bar, 20 μm. ( H ) Quantitative analysis of the capillary density in the ischemic heart. (I) Quantitative analysis of the arteriole density in the ischemic heart. ( J - M ) Protein expression of VEGF, bFGF and IGF-1 determined by Western blotting in ischemic myocardium, with GAPDH as the internal control. Values are the mean ± SEM. Significant differences were determined by using one-way ANOVA. N = 5/group. *p < 0.01 vs. MI; # p < 0.01 vs. MI-MSC-vehicle
Index in PubMed under a CC BY license. PMID: 38102643
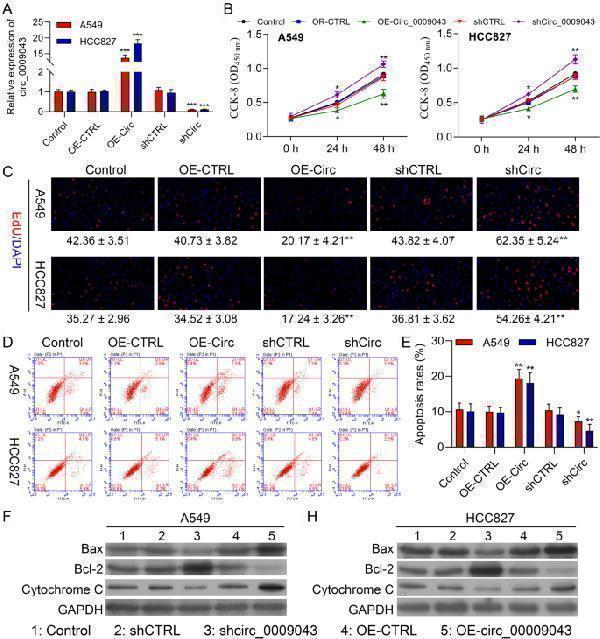
Click image to see more details
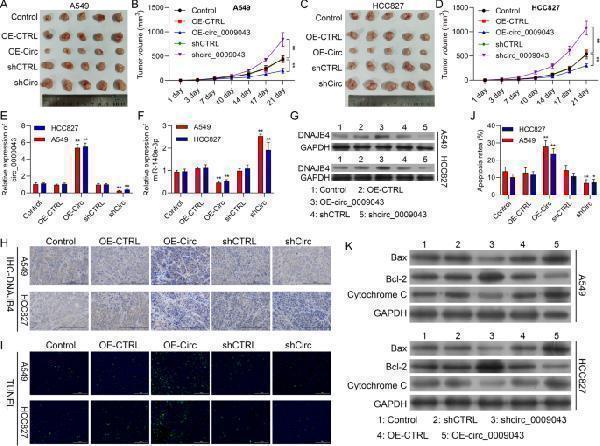
Overexpression of circ_0009043 inhibits the proliferation, while accelerates apoptosis of NSCLC cells. A circ_0009043 mRNA expression in HCC827 and A549 cells transfected with pcDNA4.0 vector (OE-CTRL), pcDNA4.0- circ_0009043 vector (OE-Circ), CTRL‑shRNA (shCTRL), or circ_0009043‑shRNA (shCirc) were determined by RT-qPCR. B CCK8 assay was used to compare the cell proliferation of Control, OE-CTRL, OE-Circ, shCTRL, and shCirc groups in HCC827 and A549 cells. C Edu assay of Control, OE-CTRL, OE-Circ, shCTRL, and shCirc groups in HCC827 and A549 cells. D-E The ratio of apoptosis in the HCC827 and A549 cells transfected with indicated vectors, which consisting of the OE-CTRL, OE- Circ, shCTRL and shCirc groups were detected by flow cytometry. Comparison of the ratio of apoptosis in the afore mentioned 5 groups. Each bar indicates the mean apoptosis rate ± standard deviation per group. F-G Expression level of Bax, Bcl-2 and Cytochrome C in the HCC827 and A549 cells of the 5 groups (CTRL, OE-CTRL, OE- Circ, shCTRL and shCirc groups) were determined by western blotting. Data was normalized to GAPDH. All results were representative of three separate experiments. Data represent mean values ± SD from three replicates of each sample; * P < 0.05, ** P < 0.01, *** P < 0.001
Index in PubMed under a CC BY license. PMID: 35974419
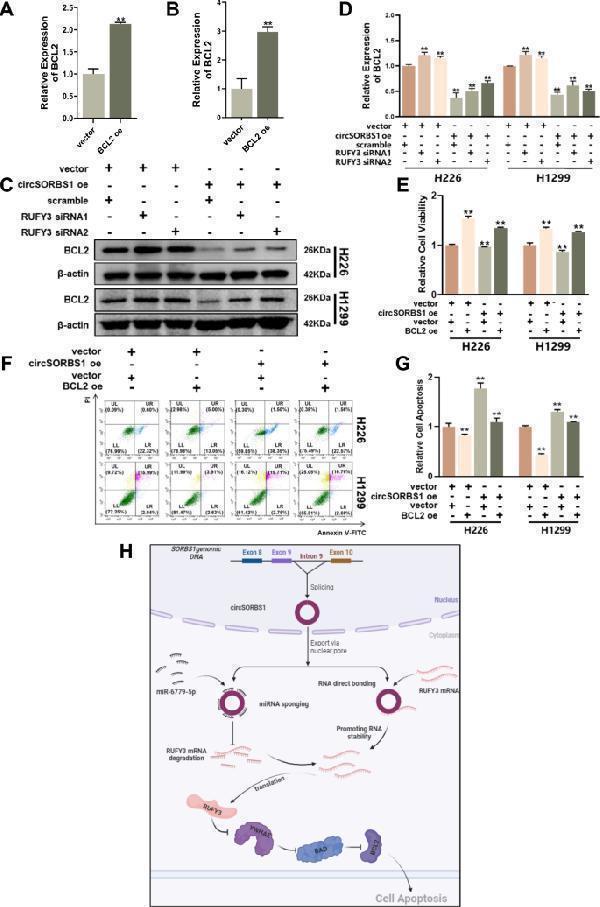
Click image to see more details
circSORBS1 inhibits lung cancer development through the RUFY3/YWHAE/BAD/BCL2 pathway. A , B qPCR detection of the transient overexpression efficiency of BCL2 mRNA in H226 versus H1299 cells. C , D . Western blot analysis of YWHAE protein expression after transient silencing and overexpression of RUFY3 mRNA and grey value analysis. E A CCK-8 assay was used to detect the viability of H1299 and H226 cells after transfection with BCL2. F , G Flow cytometry was used to detect the apoptotic capacity after BCL2 backfilling. H circSORBS1 acts as a miR-6779-5p sponge and indirectly inhibits RUFY3 mRNA degradation, directly binds to RUFY3 mRNA and enhances its stability, which in turn increases RUFY3 protein expression, activates the YWHAE/BAD/BCL2 apoptotic signalling pathway, and inhibits lung cancer progression
Index in PubMed under a CC BY license. PMID: 38915053
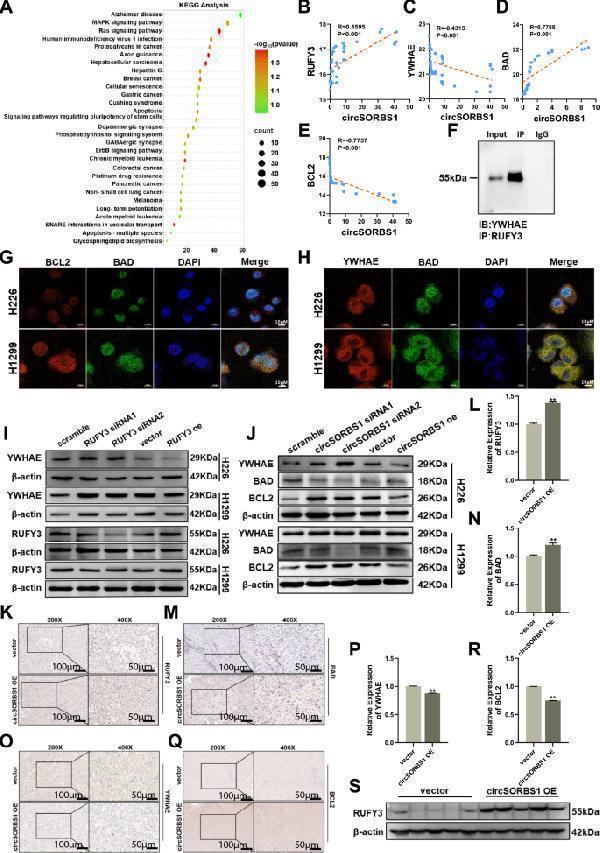
Click image to see more details
circSORBS1 regulates apoptosis via the RUFY3/YWHAE pathway. A KEGG pathway analysis of genes downstream of RUFY3. B–E Correlation analysis of circSORBS1 with RUFY3 and key proteins of the apoptotic pathways YWHAE, BAD, and BCL2 in the UCSC database. F Results of the Western blot analysis for Co-IP experiments. G IF was used to detect the colocalization of the BCL2 protein with the BAD protein. H IF was used to detect the colocalization of YWHAE with the BAD protein. I Western blot analysis of YWHAE and RUFY3 protein expression after transient RUFY3 silencing and overexpression. J Western blot analysis of YWHAE and RUFY3 protein expression after transient circSORBS1 silencing and overexpression. K–R Immunohistochemistry was performed to detect the protein expression levels of RUFY3, YWHAE, BAD, and BCL2 in nude mouse tumours. S Western blot analysis of RUFY3 protein expression in the tumour tissue of nude mice
Index in PubMed under a CC BY license. PMID: 38915053
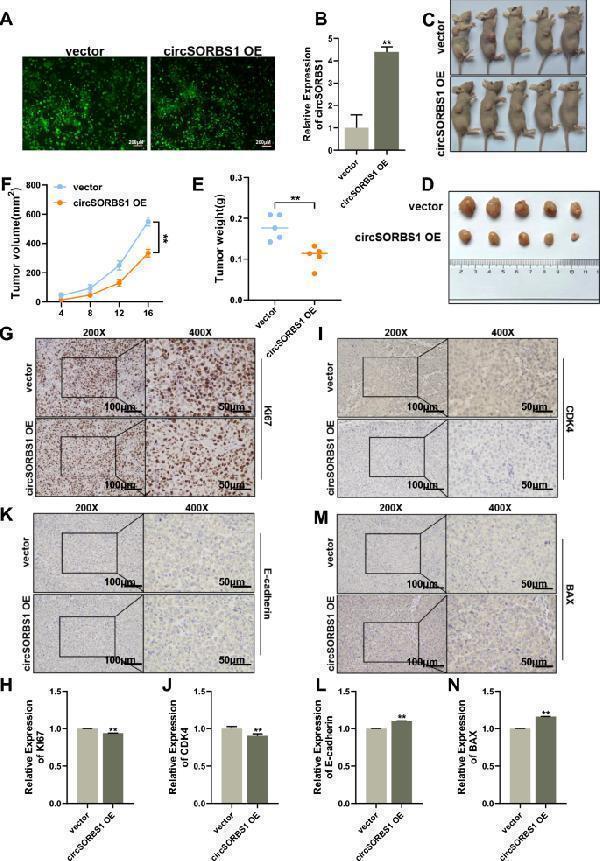
Click image to see more details
circSORBS1 inhibits lung cancer development in vivo. A Construction of a circSORBS1 stable overexpression cell line by lentiviral transfection of H226 cells. B qPCR detection of circSORBS1 overexpression efficiency in H226 cells stably overexpressing circSORBS1. C , D Nude tumour formation experiments and tumour size determination. E Statistical analysis of tumour weight in nude mice. F Growth of the nude mice. G – N Immunohistochemical analysis of the protein expression of Ki67, CDK4, BAX, and E-cadherin in nude tumours
Index in PubMed under a CC BY license. PMID: 38915053
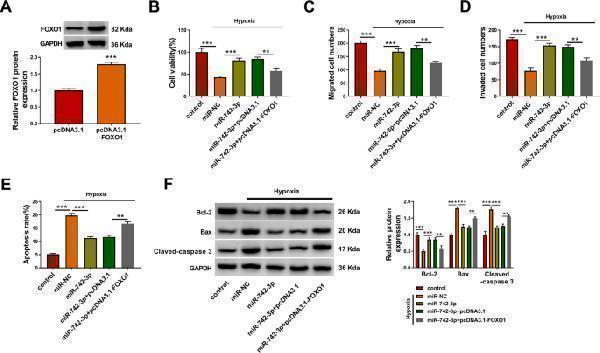
Click image to see more details
Effects of miR-742-3p and FOXO1 on hypoxia-induced H9c2 cell injury. A H9c2 cells were transfected with pcDNA3.1 and pcDNA3.1- FOXO1 , and the protein expression of FOXO1 was detected by WB analysis. B – F H9c2 cells were transfected with miR-NC (50 nM), miR-742-3p (50 nM), miR-742-3p (50 nM) + pcDNA3.1 (4.0 µg), or miR-742-3p (50 nM) + pcDNA3.1- FOXO1 (4.0 µg), and then treated with hypoxia. Untreated H9c2 cells were used as control. Cell viability, migrated and invaded cell numbers, and cell apoptosis rate were determined by CCK8 assay ( B ), transwell assay ( C , D ), and flow cytometry ( E ). F WB analysis was employed to examine the protein levels of Bcl-2, Bax and Cleaved-caspase 3. All experiments were repeated three times. ** P < 0.01, *** P < 0.001
Index in PubMed under a CC BY license. PMID: 35346026
Click image to see more details
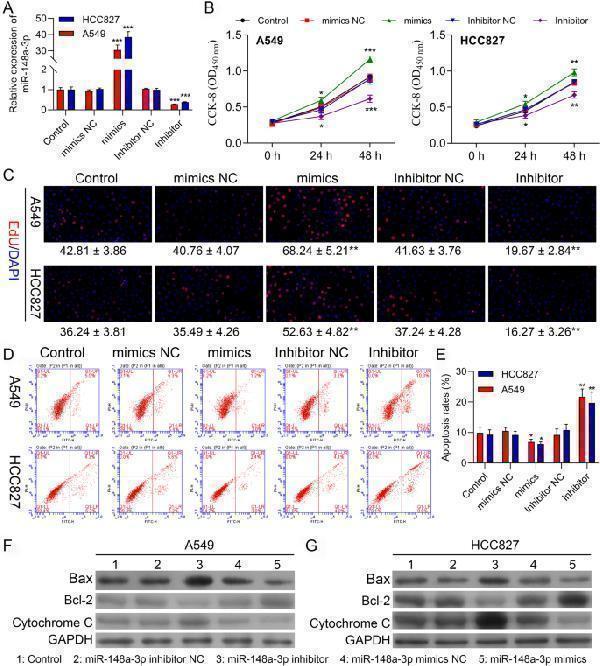
MiR-148a-3p promotes proliferation, while inhibits apoptosis in A549 and HCC827 cells. A qPCR assay confirming the transfection efficiency of the miR-148a-3p mimic and miR-148a-3p inhibitor in A549 and HCC827 cells. B Viability in A549 and HCC827 cells transfected with miR-NC, miR-148a-3p mimic, miR-148a-3p inhibitor NC, or miR-148a-3p inhibitor. C Edu assay of cell proliferation in A549 and HCC827 cells transfected with miR-NC, miR-148a-3p mimic, miR-148a-3p inhibitor NC, or miR-148a-3p inhibitor, respectively. D-E Cell apoptosis rates of A549 and HCC827 cells with the indicated transfection were determined with FSC assay. Data are presented as mean ± standard deviation. The experiments were repeated three times. F-G Protein levels of Bax, Bcl-2 and Cytochrome in A549 and HCC827 cells with the indicated transfection were determined by western blot. Data are presented as mean ± standard deviation. The experiments were repeated three times. * P < 0.05; ** P < 0.01; *** P < 0.001
Index in PubMed under a CC BY license. PMID: 35974419
Click image to see more details
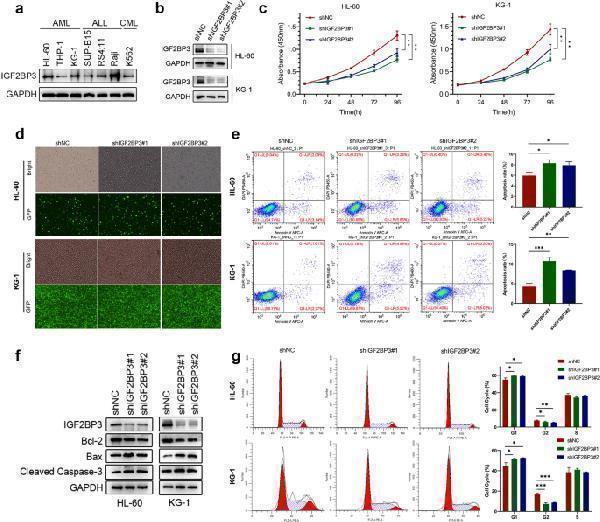
Knockdown of IGF2BP3 significantly inhibits AML progression in vitro. a The protein expression level of IGF2BP3 in various hematologic tumor cell lines was measured by western blotting. b The knockdown efficiency of IGF2BP3 shRNAs (shIGF2BP3#1 and shIGF2BP3#2) delivered via lentiviral vectors in HL-60 and KG-1 cell lines was confirmed by western blotting. GAPDH was used as the internal reference. c Cell proliferation was measured by a CCK-8 assay at different time points (0, 24, 48, 72, and 96 h) in HL-60 and KG-1 cells after shRNA transduction. d The transduction efficiency after puromycin selection was evaluated by GFP fluorescence imaging in both cell lines. e Flow cytometry (representative images are presented) was used to confirm the induction of apoptosis by IGF2BP3 knockdown. f Western blotting was used to explore apoptosis-related protein levels. The levels of cleaved caspase-3 and Bax were increased but the level of Bcl-2 was decreased under shIGF2BP3 treatment compared with control treatment. g Flow cytometry (representative images are presented) was used to analyze the cell cycle distribution. * P < 0.05; ** P < 0.01; *** P < 0.001.
Index in PubMed under a CC BY license. PMID: 35217832
Click image to see more details
Circ_0009043 inhibits tumor growth via targeting the miR-148a-3p / DNAJB4 pathway in vivo. A, C Tumors formed 6 weeks post-injection in BALB/C nude mice. Tumors in the CTRL, OE-CTRL, OE-Circ, shCTRL and shCirc groups were isolated from mice at the endpoint of experiments. B, D Tumor growth was assessed by tumor volume measurement over time in the 5 afore mentioned groups (mean ± SD; n = 5). ** P < 0.01. Mice were anesthetized and sacrificed at experimental endpoints. Tumors were subsequently dissected. E, F Circ_0009043 and miR-148a-3p mRNA expression in tumors from CTRL, OE-CTRL, OE-Circ, shCTRL and shCirc groups, respectively; n = 5. G DNAJB4 protein expression in tumors from CTRL, OE-CTRL, OE-Circ, shCTRL and shCirc groups; n = 5. H Representative images of DNAJB4 IHC in CTRL, OE-CTRL, OE-Circ, shCTRL and shCirc groups, respectively. (× 200, scale bars, 100 µm). I-J TUNEL staining assay was applied to compare the cell apoptosis of CTRL, OE-CTRL, OE-Circ, shCTRL and shCirc groups in tumors (scale bar, 100 μm). K Expression level of Bax, Bcl-2, and Cytochrome C in the cells of the 5 groups (CTRL, OE-CTRL, OE-Circ, shCTRL and shCirc groups) were determined by western blotting. ** P < 0.01, *** P < 0.001, ### P < 0.001; #### P < 0.0001
Index in PubMed under a CC BY license. PMID: 35974419
Specific Publications For Anti-Apoptosis regulator BAX Bax Antibody Picoband® (PA1013-1)
Loading publications
Recommended Resources
Here are featured tools and databases that you might find useful.
- Boster's Pathways Library
- Protein Databases
- Bioscience Research Protocol Resources
- Data Processing & Analysis Software
- Photo Editing Software
- Scientific Literature Resources
- Research Paper Management Tools
- Molecular Biology Software
- Primer Design Tools
- Bioinformatics Tools
- Phylogenetic Tree Analysis
Customer Reviews
Have you used Anti-Apoptosis regulator BAX Bax Antibody Picoband®?
Share your experimental results or join a short interview to earn up to $1,000 in product credits or other rewards.
0 Reviews For Anti-Apoptosis regulator BAX Bax Antibody Picoband®
Customer Q&As
Have a question?
Find answers in Q&As, reviews.
Can't find your answer?
Submit your question
17 Customer Q&As for Anti-Apoptosis regulator BAX Bax Antibody Picoband®
Question
Is a blocking peptide available for product anti-Bax antibody (PA1013-1)?
Verified Customer
Verified customer
Asked: 2020-04-23
Answer
We do provide the blocking peptide for product anti-Bax antibody (PA1013-1). If you would like to place an order for it please contact support@bosterbio.com and make a special request.
Boster Scientific Support
Answered: 2020-04-23
Question
Is this PA1013-1 anti-Bax antibody reactive to the isotypes of BAX?
Verified Customer
Verified customer
Asked: 2020-04-13
Answer
The immunogen of PA1013-1 anti-Bax antibody is A synthetic peptide corresponding to a sequence at the N-terminus of human Bax(19-33aa IMKTGALLLQGFIQD), different from the related mouse and rat sequences by one amino acid. Could you tell me which isotype you are interested in so I can help see if the immunogen is part of this isotype?
Boster Scientific Support
Answered: 2020-04-13
Question
Will PA1013-1 anti-Bax antibody work on parafin embedded sections? If so, which fixation method do you recommend we use (PFA, paraformaldehyde, other)?
Verified Customer
Verified customer
Asked: 2020-02-28
Answer
It shows on the product datasheet, PA1013-1 anti-Bax antibody as been validated on WB. It is best to use PFA for fixation because it has better tissue penetration ability. PFA needs to be prepared fresh before use. Long term stored PFA turns into formalin, as the PFA molecules congregate and become formalin.
Boster Scientific Support
Answered: 2020-02-28
Question
I am interested in using your anti-Bax antibody for post-embryonic camera-type eye morphogenesis studies. Has this antibody been tested with western blotting on tissue lysate? We would like to see some validation images before ordering.
Verified Customer
Verified customer
Asked: 2019-10-10
Answer
We appreciate your inquiry. This PA1013-1 anti-Bax antibody is validated on rat ovary tissue, testis tissue, tissue lysate, kidney tissue, brain tissue, hela cell lysate, mm231 cell lysate, a549 cell lysate, jurkat cell lysate, human placenta tissue, a431 cells. It is guaranteed to work for Flow Cytometry, WB in human, mouse, rat. Our Boster guarantee will cover your intended experiment even if the sample type has not been be directly tested.
Boster Scientific Support
Answered: 2019-10-10
Question
Thank you for helping with my inquiry over the phone. Here are the WB image, lot number and protocol we used for mucosa of transverse colon using anti-Bax antibody PA1013-1. Let me know if you need anything else.
Verified Customer
Verified customer
Asked: 2019-09-16
Answer
We appreciate the data. You have provided everything we needed. Our lab team are working to resolve your inquiry as quickly as possible, and we appreciate your patience and understanding! Please let me know if there is anything you need in the meantime.
Boster Scientific Support
Answered: 2019-09-16
Question
Can you help my question with product PA1013-1, anti-Bax antibody. I was wondering if it would be possible to conjugate this antibody with biotin. I would need it to be without BSA or sodium azide. I am planning on using a buffer exchange of sodium azide with PBS only. Would there be problems for me to conjugate the antibody and store it in -20 degrees in small aliquots?
Verified Customer
Verified customer
Asked: 2019-08-19
Answer
We do not advise storing this antibody with PBS buffer only in -20 degrees. If you want to store it in -20 degrees it is best to add some cryoprotectant like glycerol. If you want carrier free PA1013-1 anti-Bax antibody, we can provide it to you in a special formula with trehalose and/or glycerol. These molecules will not interfere with conjugation chemistry and provide a good level of protection for the antibody from degradation. Please be sure to specify this in your purchase order.
Boster Scientific Support
Answered: 2019-08-19
Question
Does anti-Bax antibody PA1013-1 work on monkey WB with brain?
Verified Customer
Verified customer
Asked: 2019-07-19
Answer
Our lab technicians have not tested anti-Bax antibody PA1013-1 on monkey. You can run a BLAST between monkey and the immunogen sequence of anti-Bax antibody PA1013-1 to see if they may cross-react. If the sequence homology is close, then you can perform a pilot test. Keep in mind that since we have not validated monkey samples, this use of the antibody is not covered by our guarantee. However we have an innovator award program that if you test this antibody and show it works in monkey brain in WB, you can get your next antibody for free.
Boster Scientific Support
Answered: 2019-07-19
Question
Is there a BSA free version of anti-Bax antibody PA1013-1 available?
Verified Customer
Verified customer
Asked: 2019-06-14
Answer
We appreciate your recent telephone inquiry. I can confirm that some lots of this anti-Bax antibody PA1013-1 are BSA free. For now, these lots are available and we can make a BSA free formula for you free of charge. It will take 3 extra days to prepare. If you require this antibody BSA free again in future, please do not hesitate to contact me and I will be pleased to check which lots we have in stock that are BSA free.
Boster Scientific Support
Answered: 2019-06-14
Question
We are interested in to test anti-Bax antibody PA1013-1 on human mucosa of transverse colon for research purposes, then I may be interested in using anti-Bax antibody PA1013-1 for diagnostic purposes as well. Is the antibody suitable for diagnostic purposes?
Verified Customer
Verified customer
Asked: 2018-12-21
Answer
The products we sell, including anti-Bax antibody PA1013-1, are only intended for research use. They would not be suitable for use in diagnostic work. If you have the means to develop a product into diagnostic use, and are interested in collaborating with us and develop our product into an IVD product, please contact us for more discussions.
Boster Scientific Support
Answered: 2018-12-21
Question
We bought anti-Bax antibody for Flow Cytometry on mucosa of transverse colon a few years ago. I am using human, and We intend to use the antibody for WB next. I was wanting to use examining mucosa of transverse colon as well as ovarian carcinoma in our next experiment. Do you have any suggestion on which antibody would work the best for WB?
P. Dhar
Verified customer
Asked: 2018-06-12
Answer
I looked at the website and datasheets of our anti-Bax antibody and it appears that PA1013-1 has been validated on human in both Flow Cytometry and WB. Thus PA1013-1 should work for your application. Our Boster satisfaction guarantee will cover this product for WB in human even if the specific tissue type has not been validated. We do have a comprehensive range of products for WB detection and you can check out our website bosterbio.com to find out more information about them.
Boster Scientific Support
Answered: 2018-06-12
Question
We have been able to see staining in mouse skin. Do you have any suggestions? Is anti-Bax antibody supposed to stain skin positively?
Verified Customer
Verified customer
Asked: 2018-03-22
Answer
From literature skin does express BAX. From Uniprot.org, BAX is expressed in mucosa of transverse colon, b-cell, brain, ovarian carcinoma, skin, among other tissues. Regarding which tissues have BAX expression, here are a few articles citing expression in various tissues:
B-cell, Pubmed ID: 8358790
Brain, Pubmed ID: 9920818
Ovarian carcinoma, Pubmed ID: 14702039
Skin, Pubmed ID: 15489334
Boster Scientific Support
Answered: 2018-03-22
Question
My colleagues were content with the WB result of your anti-Bax antibody. However we have seen positive staining in skin isoform beta: cytoplasm. using this antibody. Is that expected? Could you tell me where is BAX supposed to be expressed?
K. Johnson
Verified customer
Asked: 2017-11-21
Answer
According to literature, skin does express BAX. Generally BAX expresses in isoform alpha: mitochondrion outer membrane, isoform beta: cytoplasm., isoform gamma: cytoplasm., isoform delta: cytoplasm. Regarding which tissues have BAX expression, here are a few articles citing expression in various tissues:
B-cell, Pubmed ID: 8358790
Brain, Pubmed ID: 9920818
Ovarian carcinoma, Pubmed ID: 14702039
Skin, Pubmed ID: 15489334
Boster Scientific Support
Answered: 2017-11-21
Question
I have attached the WB image, lot number and protocol we used for mucosa of transverse colon using anti-Bax antibody PA1013-1. Please let me know if you require anything else.
Verified Customer
Verified customer
Asked: 2017-09-22
Answer
Thank you very much for the data. Our lab team are working to resolve this as quickly as possible, and we appreciate your patience and understanding! You have provided everything we needed. Please let me know if there is anything you need in the meantime.
Boster Scientific Support
Answered: 2017-09-22
Question
I was wanting to use your anti-Bax antibody for WB for human mucosa of transverse colon on frozen tissues, but I want to know if it has been validated for this particular application. Has this antibody been validated and is this antibody a good choice for human mucosa of transverse colon identification?
K. Kulkarni
Verified customer
Asked: 2017-02-16
Answer
You can see on the product datasheet, PA1013-1 anti-Bax antibody has been validated for Flow Cytometry, WB on human, mouse, rat tissues. We have an innovator award program that if you test this antibody and show it works in human mucosa of transverse colon in IHC-frozen, you can get your next antibody for free.
Boster Scientific Support
Answered: 2017-02-16
Question
I see that the anti-Bax antibody PA1013-1 works with WB, what is the protocol used to produce the result images on the product page?
A. Jackson
Verified customer
Asked: 2016-09-09
Answer
You can find protocols for WB on the "support/technical resources" section of our navigation menu. If you have any further questions, please send an email to support@bosterbio.com
Boster Scientific Support
Answered: 2016-09-09
Question
Does anti-Bax antibody PA1013-1 work for WB with mucosa of transverse colon?
G. Krishna
Verified customer
Asked: 2015-06-02
Answer
According to the expression profile of mucosa of transverse colon, BAX is highly expressed in mucosa of transverse colon. So, it is likely that anti-Bax antibody PA1013-1 will work for WB with mucosa of transverse colon.
Boster Scientific Support
Answered: 2015-06-02
Question
We are currently using anti-Bax antibody PA1013-1 for mouse tissue, and we are well pleased with the WB results. The species of reactivity given in the datasheet says human, mouse, rat. Is it true that the antibody can work on monkey tissues as well?
D. Carter
Verified customer
Asked: 2015-02-02
Answer
The anti-Bax antibody (PA1013-1) has not been validated for cross reactivity specifically with monkey tissues, though there is a good chance of cross reactivity. We have an innovator award program that if you test this antibody and show it works in monkey you can get your next antibody for free. Please contact me if I can help you with anything.
Boster Scientific Support
Answered: 2015-02-02





