This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
and ELISA kits, proteins related to Apoptosis.
Apoptosis, often referred to as programmed cell death, is a vital biological process that maintains cellular balance and overall organism health. Unlike accidental cell death, apoptosis is a highly regulated and orderly mechanism that eliminates damaged, unnecessary, or potentially harmful cells without triggering inflammation. This process is essential for various physiological functions, including embryonic development, immune system regulation, and tissue homeostasis. Dysregulation of apoptosis is closely linked to numerous diseases, such as cancer, autoimmune disorders, and neurodegenerative conditions, highlighting its significance in medical research. Advanced research tools, particularly apoptosis-related antibodies, play a crucial role in unraveling the complex pathways of cell death. These antibodies enable scientists to detect, quantify, and manipulate specific proteins involved in apoptosis, thereby facilitating breakthroughs in understanding and treating a wide range of health issues.
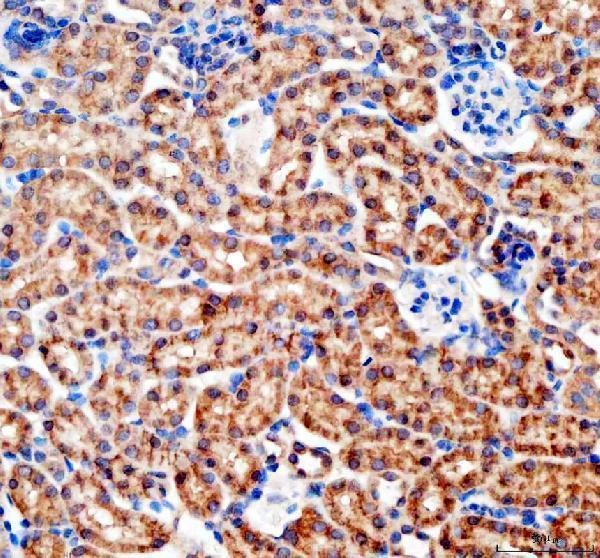
Anti-Bax Antibody Picoband®, Figure 3. IHC analysis of Bax using anti-Bax antibody (A00183).
Bax was detected in paraffin-embedded section of rat intestine tissues. Heat media...
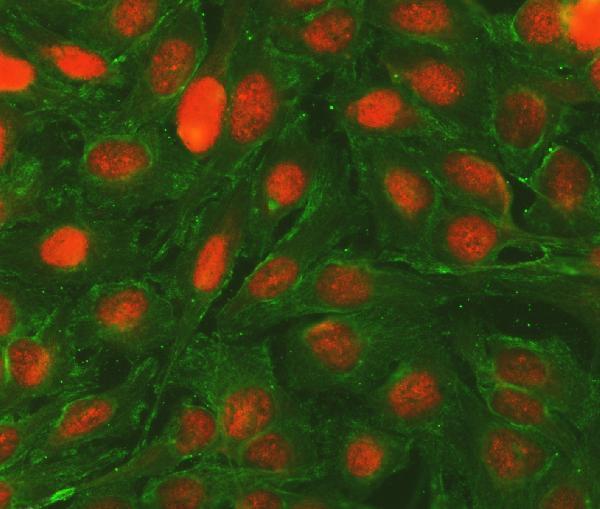
Anti-Bcl-2/BCL2 Antibody Picoband®, Figure 2. IF analysis of BCL2 and Tubulin alpha using anti-BCL2 antibody (A00040-2) and anti-Tubulin alpha antibody (M03989-3).
BCL2 a...
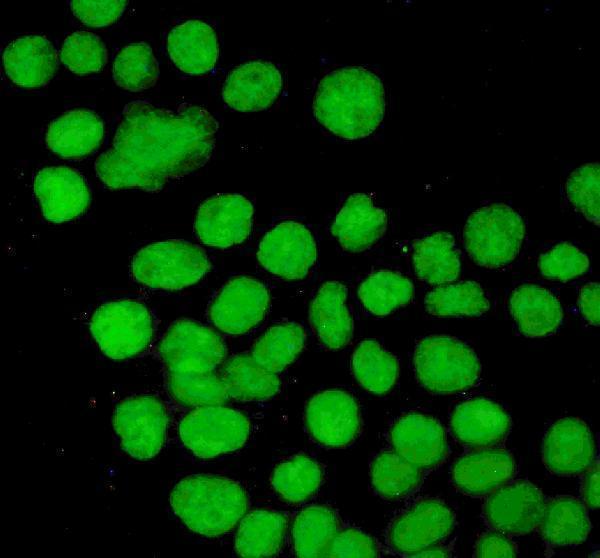
Anti-P53/TP53 Antibody Picoband®, Figure 3. IF analysis of P53 using anti-P53 antibody (PB9008).
P53 was detected in immunocytochemical section of A431 cells. Enzyme antigen...
| Protein Name | Gene Name | Function |
|---|---|---|
| Caspase-3 | CASP3 | Executioner caspase that carries out apoptosis by cleaving cellular substrates. |
| Bcl-2 | BCL2 | Anti-apoptotic protein that inhibits mitochondrial outer membrane permeabilization. |
| Bax | BAX | Pro-apoptotic protein that promotes mitochondrial membrane permeabilization. |
| Cytochrome c | CYCS | Mediates apoptosome formation leading to caspase activation. |
| p53 | TP53 | Tumor suppressor that induces apoptosis in response to DNA damage. |
| Apaf-1 | APAF1 | Apoptotic protease activating factor that forms the apoptosome with cytochrome c. |
| Caspase-8 | CASP8 | Initiator caspase in the extrinsic apoptotic pathway. |
| Caspase-9 | CASP9 | Initiator caspase in the intrinsic apoptotic pathway. |
| BIM | BCL2L11 | Pro-apoptotic BH3-only protein that activates Bax and Bak. |
| BAD | BAD | Pro-apoptotic BH3-only protein that inhibits Bcl-2 and Bcl-xL. |
| Bid | BID | BH3-only protein that links extrinsic and intrinsic apoptotic pathways. |
| XIAP | XIAP | Inhibitor of apoptosis proteins that block caspase activity. |
| Smac/DIABLO | DIABLO | Promotes apoptosis by antagonizing IAPs and facilitating caspase activation. |
| FADD | FADD | Adaptor molecule that transmits apoptotic signals from death receptors. |
| TRAIL | TNFSF10 | Ligand that induces apoptosis by binding to death receptors DR4 and DR5. |
| Fas ligand | FASLG | Protein that binds Fas receptor to trigger the extrinsic apoptotic pathway. |
| Mcl-1 | MCL1 | Anti-apoptotic Bcl-2 family protein that regulates mitochondrial integrity. |
| Noxa | PMAIP1 | BH3-only protein that selectively antagonizes Mcl-1. |
| Puma | BBC3 | BH3-only protein that promotes apoptosis by inhibiting anti-apoptotic Bcl-2 family members. |
The intrinsic pathway of apoptosis, also known as the mitochondrial pathway, is a pivotal mechanism of programmed cell death that is primarily regulated by intracellular signals in response to cellular stress, DNA damage, or other internal stimuli. Central to this pathway is the mitochondria, which act as key regulators by releasing pro-apoptotic factors such as cytochrome c into the cytoplasm upon receiving apoptotic signals. Once released, cytochrome c interacts with Apaf-1 and procaspase-9 to form the apoptosome, a multi-protein complex that activates caspase-9. Activated caspase-9 then initiates a cascade of downstream effector caspases, like caspase-3, leading to the systematic dismantling of cellular components and eventual cell death. The intrinsic pathway is tightly controlled by the Bcl-2 family of proteins, which include both pro-apoptotic and anti-apoptotic members. The balance between these opposing forces determines the cell’s fate, ensuring that apoptosis is executed accurately in response to genuine cellular threats. Understanding the intricacies of the intrinsic pathway is crucial for developing therapeutic strategies aimed at diseases where apoptosis is dysregulated, such as cancer and neurodegenerative disorders.
The extrinsic pathway of apoptosis is initiated by extracellular signals that bind to specific death receptors on the cell surface, triggering a cascade of events leading to programmed cell death. Key players in this pathway include receptors such as Fas (CD95) and the TNF-related apoptosis-inducing ligand (TRAIL) receptors. Upon ligand binding, these death receptors undergo conformational changes that recruit adaptor proteins like FADD (Fas-associated death domain) and procaspase-8 to form the death-inducing signaling complex (DISC). Within the DISC, procaspase-8 is activated to caspase-8, which then directly activates downstream effector caspases, including caspase-3, culminating in apoptosis. Additionally, caspase-8 can cleave the BH3-only protein Bid, linking the extrinsic pathway to the intrinsic mitochondrial pathway and amplifying the apoptotic signal. The extrinsic pathway plays a vital role in maintaining tissue homeostasis, immune system regulation, and the removal of harmful or infected cells. Dysregulation of this pathway is implicated in various pathologies, including autoimmune diseases, chronic inflammation, and cancer, making it a significant focus of research for therapeutic intervention.